Your Cart is Empty
The Beauty of Black Rings: Black Rhodium vs Oxidised Silver
Thinking of going bold with black rings? Here’s what you need to know!
For gothic brides and divas out there looking for that slightly misbehaving look, a black ring shouldn’t be too much to ask.
In fact, black metal can set a nice contrast to your gemstone. It can make the sparkle of your gem more radiant and luminous. What’s not to love, right?
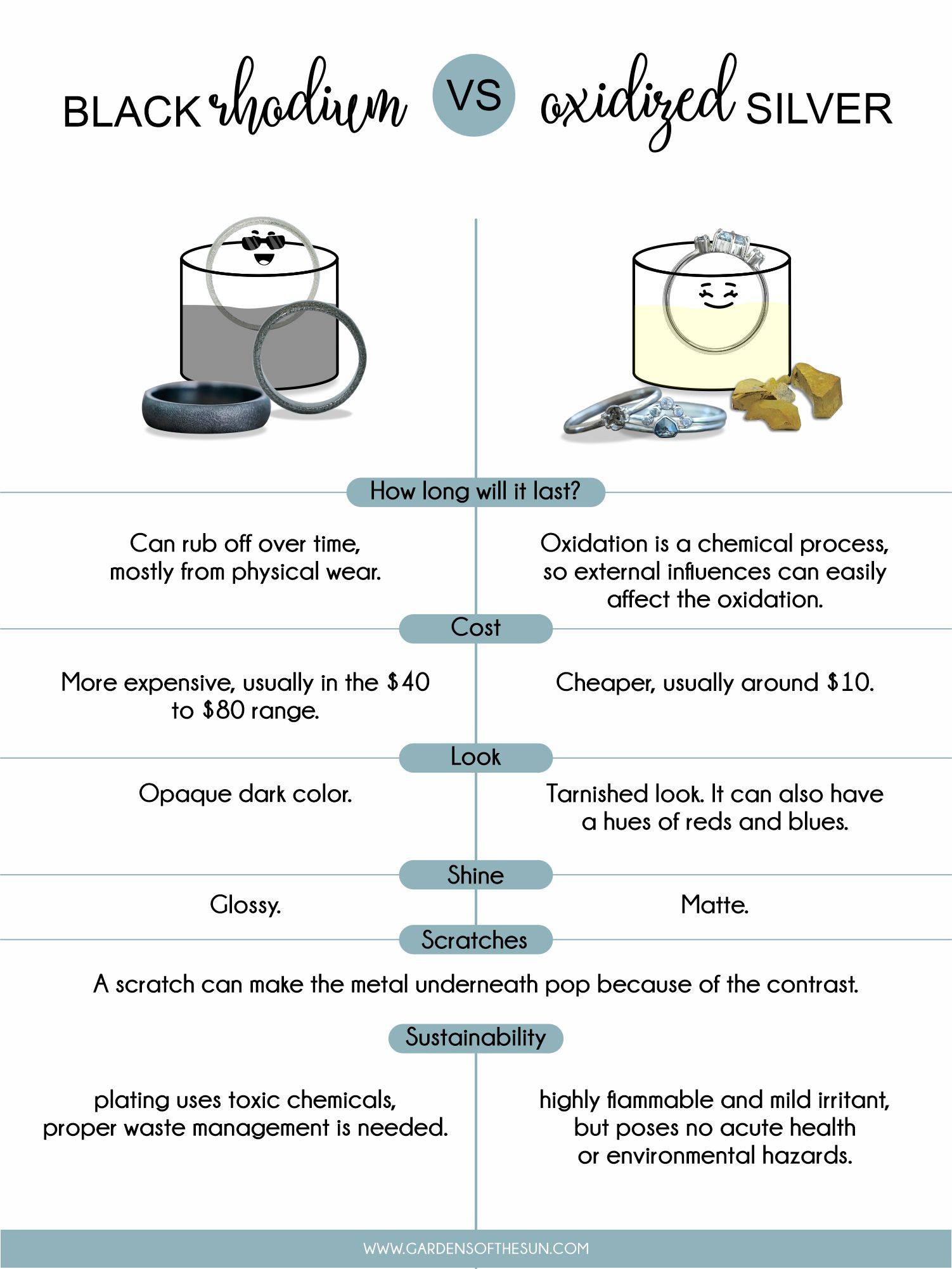
So there are two ways to turn your jewelry black: black rhodium and oxidized silver. They’re different processes for coloring the surface layer of your metal, and they yield different results.
So what are they and which one is right for you?
Black Rhodium
A shimmering black rhodium plated ring. Would you go plain or bold with a colorful gemstone?
Let’s go back to rhodium plating for a sec.
Rhodium is a rare metal which is highly reflective and doesn’t tarnish, but is too brittle on its own. When used as a plating, rhodium increases the metal’s durability and luster. You’d see it used on white gold ring to enhance its silvery shine, while adding a protective layer.
So is rhodium a durability-enhancer or a color treatment?
It's a color enhancer first and foremost. Rhodium’s natural color is white metal, but ink can bind to the rhodium particles and so you can dress up your jewelry in various colors.
For rhodium plating, a positive electrical charge is used to fuse the rhodium onto the base metal. Within minutes, your silver or gold jewelry will turn black.
So if you use black rhodium, do you get the best out of both worlds?
Wearability
Here’s the thing. Black rhodium is a surface-color treatment. Right underneath the thin black layer is your precious silver or gold. Any scratch can make the metal underneath pop because of the contrast.
Keep in mind that black rhodium isn’t an alloy. It’s not like a rose gold ring, where the colors are made from a mixture of metals. Black rhodium isn’t the base metal, it’s a plating. Even though rhodium itself is relatively scratch-resistant, sooner or later, the color will fade and need to be re-dipped.
THE LOOK
Gothic and slightly misbehaving
Nice way to make gemstones pop
Looks awesome on textured rings since black rhodium clings better to textures
Can give you an opaque dark color
Glossy
Oxidized silver
Oxidised silver looks right at home on these textured wedding rings (on the left side is turtle textured band and on the right side is shadow textured band)!
Oxidized silver is not a technique to color the jewelry per se, but a technique to speed up the ‘oxidation’ process that naturally occurs on the metal.
The result is an antique and tarnished finish; its silver color faded, revealing an uneven dark hue. People are loving it for its aged, pre-loved feel. So what does oxidation do to silver?
First thing: it’s actually not oxygen doing all the work, but sulfides, like the yellow part of the boiled egg. So yes, it’s a misnomer. Common Internet knowledge says that if you put your ring inside a bag with boiled egg cut in two, within a few hours your ring will tarnish.
Jewelers will use liver of sulphur or potassium sulfide to color its surface. Oxidation is relatively simple manual work and controlling the level of oxidation doesn’t require elaborate machinery, just good ventilation. Oxidizing involves a chemical reaction that oxidizes the metal surface and forms sulfide compounds. When you use liver of sulfur on silver, the sulfur reacts with the surface and the atmosphere to produce the grays and blacks.
Oxidation is a chemical process, so swimming in pools, handling chemicals, using (household) cleaning agents and even the composition of your sebum can affect the oxidation on your jewelry.
So is there a difference between the natural tarnishing process and the chemical one?
Not really. Both of them are only surface-level changes (not like an alloy), and can be restored in an instant. To remove the oxidation, a jeweler can polish your ring, you can use a steam cleaner or you can opt for professional cleaning. Professional cleaning can either polish the oxidized layer off, or use chemicals and steam to reverse the process, and restore its silvery shine.
Wearability
Your silver piece will continue its tarnishing process naturally. Humidity and pollution definitely play a role, as does the level of sebum in your skin. A friend of mine can't wear oxidized silver simply because the sebum on his skin clears the oxidised layer in no time. All his silver jewelry is always shiny - it’s like his skin naturally cleans silver. So understand the characteristics of your skin as well.
If you live in areas with high humidity and high pollution, your jewelry will tarnish faster. Harsh chemicals, including perfumes and hairspray also speed up the reaction process.
THE LOOK
Tarnished, antique
Has a more organic, natural vibe
Matte
Here’s a quick chart to help you compare which treatment works best for you.
If you’re wondering how black rings look on textured rings, look no further!
Designing a black ring can be tricky and both treatments come at additional cost. So be sure to email our Customer Happiness Rockstars before deciding on your black jewelry.



















